This page is only intended for products sold and distributed in the US. For ordering products, please contact our local US distributor, Bonalive Inc.
Concerning products and solutions sold in other countries, please visit international website. Please note that the products presented under International website are not FDA Cleared for US commercialization.

Indications for use
Bonalive Orthopedics granules is an implant intended to fill bony voids or gaps of the skeletal system (i.e., extremities and pelvis). These osseous defects may be the result of benign bone cysts and tumors (in adults and pediatric patients ≥ 6 years old), are surgically created or the result of traumatic injury to the bone and are not intrinsic to the stability of the bony structure. Bonalive Orthopedics granules resorbs and is replaced with bone during the healing process.
Contraindications
Bonalive Orthopedics granules should not be applied:
- To replace structures that are subject to strong mechanical stress;
- In patients that have received or are to receive chemo-therapy or radiation therapy at or near the implant site; or
- In patients with known allergy to bioactive glass.
The S53P4 bioactive glass gradually resorbs over time and is replaced by bone. The bioactive glass in the Bonalive Orthopedics granules is radiodense thus enabling postoperative radiologic evaluation.
Bonalive Orthopedics granules is available in different granule and unit sizes.
Complications
Possible complications are the same as to be expected of autogenous bone grafting procedures and general complications that may arise from anesthesia and/or surgery.
See IFU within the product package for complete information of Bonalive Orthopedics granules.
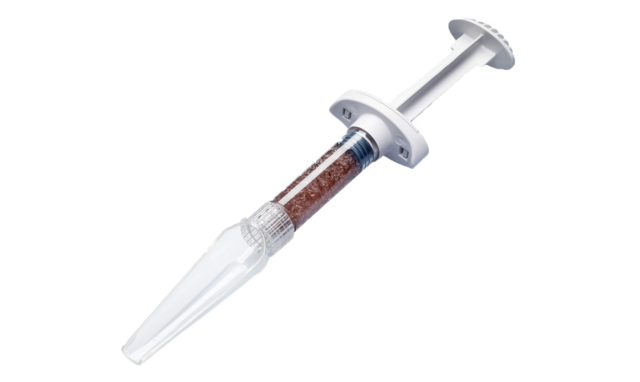
Bonalive Orthopedics granules is a sterile medical device made of S53P4 bioactive glass. This synthetic, osteoconductive material is comprised of SiO2, Na2O, CaO and P2O5. Bioactive glass is characterized by its ability to attach firmly to living tissue. Other properties include its ability to facilitate bone tissue growth, bond chemically with surrounding bone, and promote new bone formation in the implanted area.
Bonalive Orthopedics granules is only intended to be used by physicians familiar with bone grafting and fixation techniques.
- Bonalive Orthopedics granules is only intended to be used by physicians familiar with bone grafting and fixation techniques.
- Bonalive Orthopedics granules does not provide mechanical strength to support load-bearing defects before hard tissue has formed. If a fracture requires load supporting fixation, standard internal or external stabilization techniques must be used to achieve rigid stabilization in all planes.
- Screw purchase or screw stabilization must not be attempted on recent Bonalive Orthopedics granules implantation sites where sufficient bone growth is not confirmed.
- Bonalive Orthopedics granules are not intended for use with defects other than those listed in the indications for use statement. Pre-operative and post-operative patient management should follow the same regimen as similar cases utilizing autogenous bone grafting.
- The device has not been clinically tested for use in pregnant women.