We offer osteostimulative* technologies that provide an extensive range of benefits for bone regeneration.
Bonalive® granules and Bonalive® putty are highly effective technologies for the filling, reconstruction and regeneration of bone defects. Bonalive® granules also have the unique feature of naturally inhibiting bacterial growth, making it a smarter and more sustainable solution for bone regeneration.
Bonalive® putty is also available as Bonalive® putty MIS – a surgical applicator for allowing the accurate delivery of the putty to challenging voids or gaps in bone.
Please note that the country-specific marketing authorization may vary.
*non-osteoinductive
Bonalive®
granules
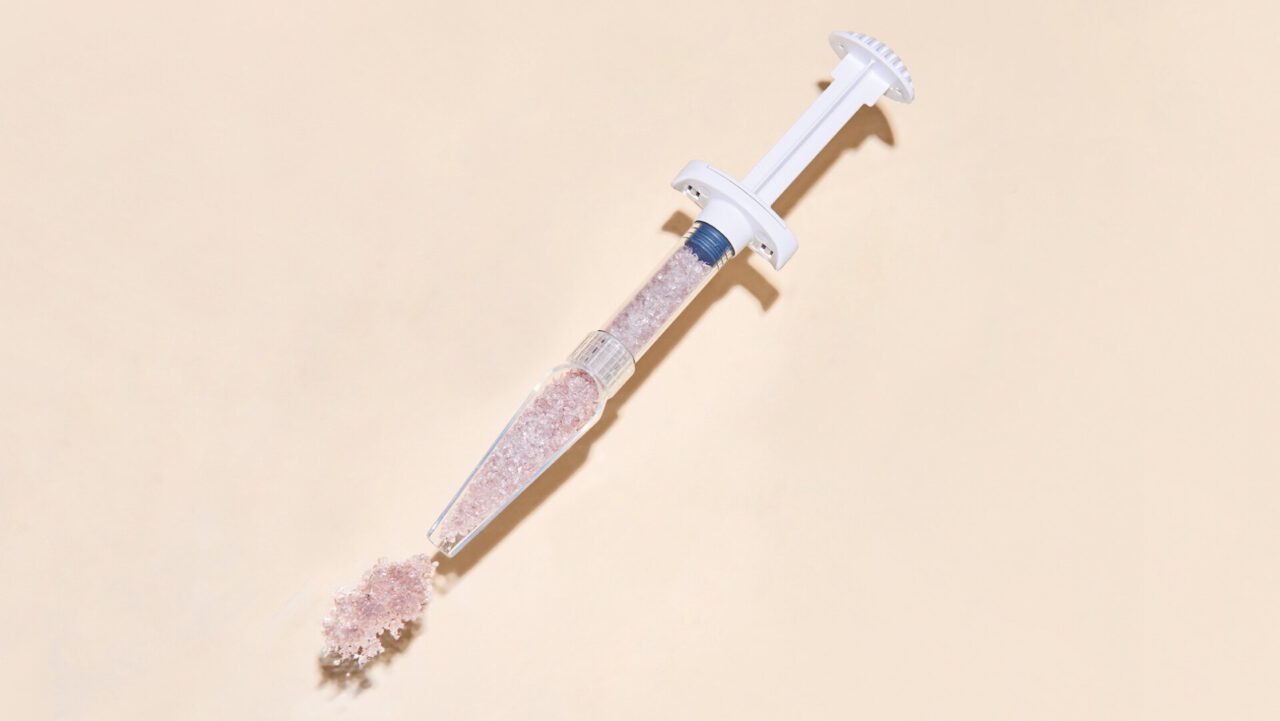
Bonalive®
granules CMF
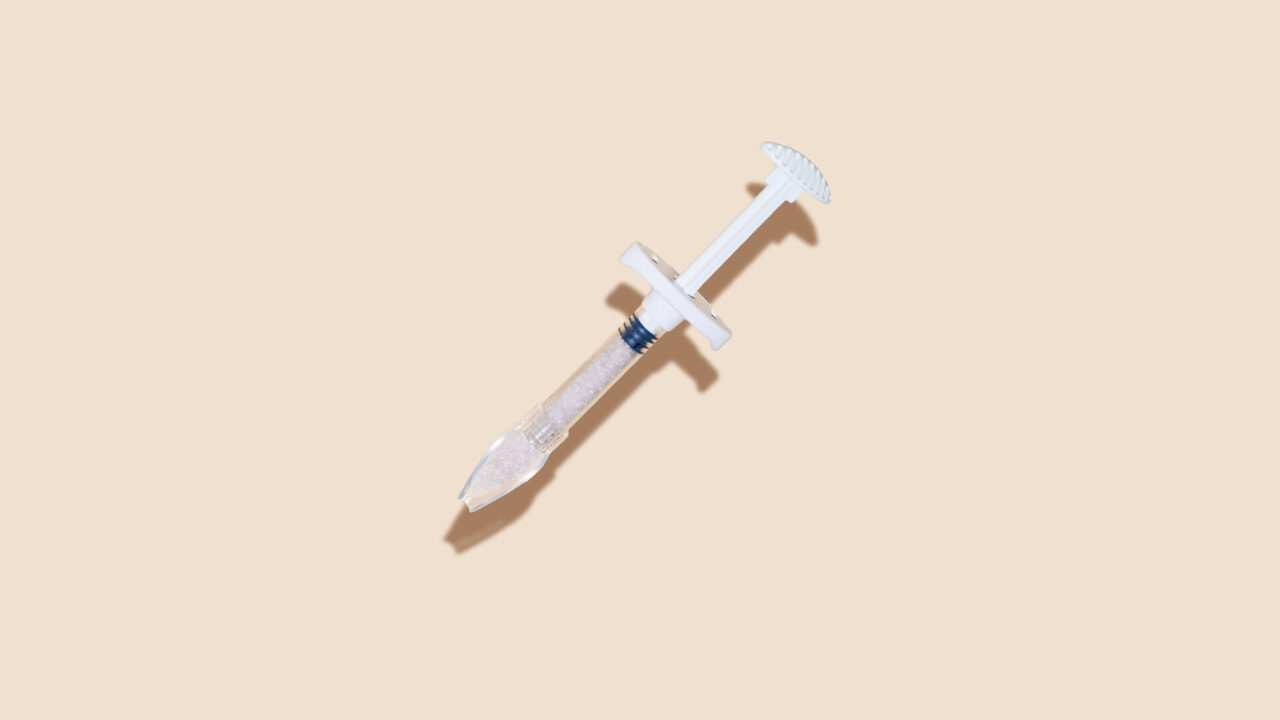
Bonalive®
putty
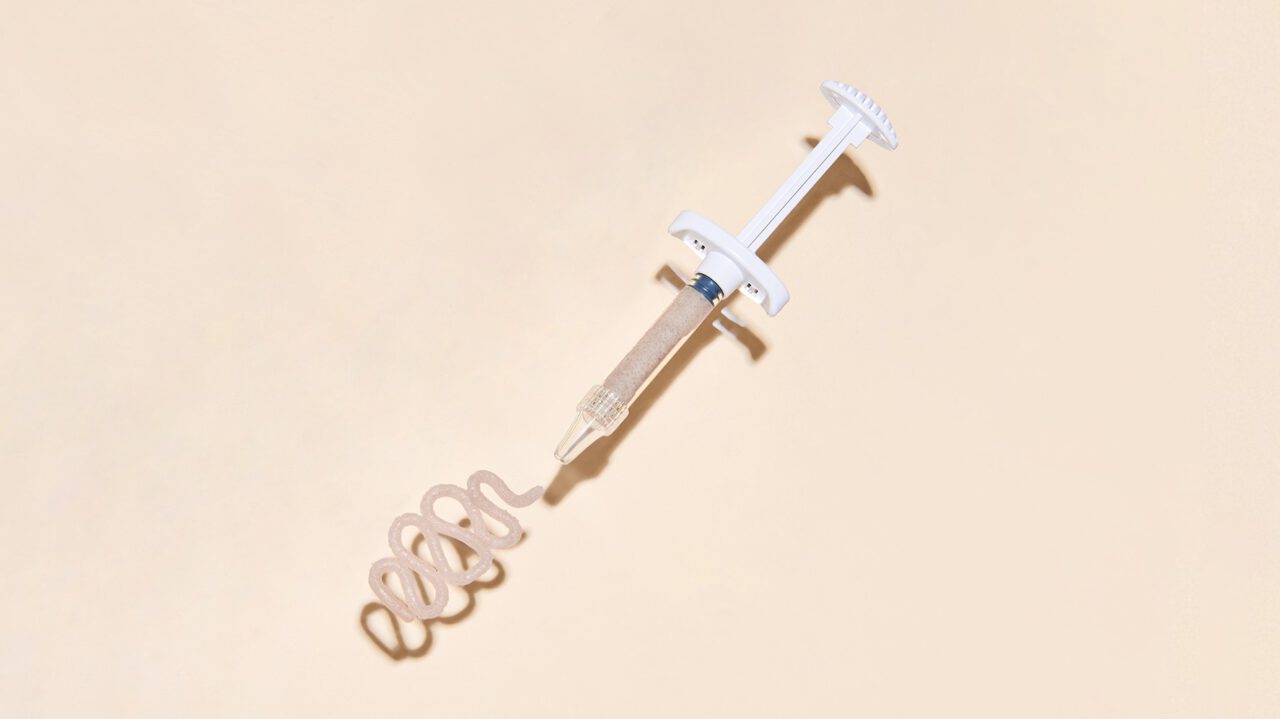
Bonalive®
putty MIS
