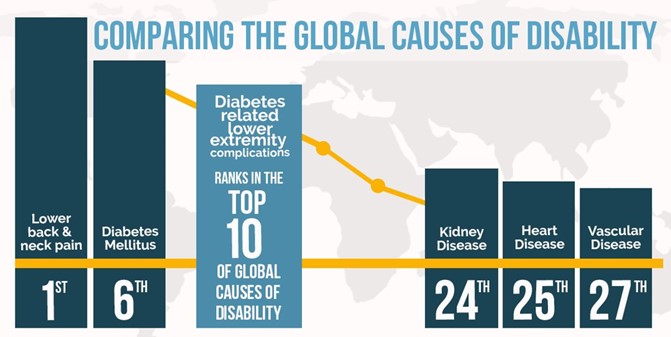
Diabetic foot infections: Why it’s important to find new sustainable solutions
The combined effect of Antimicrobial Resistance (AMR), with the projection that diabetes will increase with 40% from 500 million to 700 million people by 20451, will increase the need for improved diabetic foot infection treatments. These trends will drive the development of antibiotic free solutions to overcome the clinical issues that AMR is causing for patients with diabetic foot infections. In this blog post Bonalive’s Founder and Chairman Dr. Fredrik Ollila shares his thoughts on what kind of solutions will be needed in the future.

Societal impact of diabetic foot infection
Antimicrobial resistance is one of the top 10 global public health threats facing humanity2. Infection treatment is getting more difficult due to growing antimicrobial resistance (AMR). Every year, 2 million people with diabetes get bone infections and 1.7 million patients undergo amputation. According to specialists, 80-85% of all amputations caused by diabetes are largely preventable 3, 4, 5.
1 out of 2 patients die within 5 years after amputation6. Novel antibacterial solutions can prevent these amputations. Therefore, we have a societal responsibility to rethink how infections are treated and find new ways to outsmart bacteria.
In an interview with the Director of the Diabetic Foot Section of the Pisa University Hospital, Prof. Alberto Piaggesi stated that there’s a need to develop new treatments for infections since antibiotics lose their effectiveness. Prof. Piaggesi continued saying that especially patients with diabetes, who have a weak immune system, are particularly prone to infections, and the treatment of wounds in their lower extremities is more difficult. “We have started using Bonalive’s products at our clinic and this new technology has enabled us to save many limbs, even among young patients”, he concluded. (Helsingin Sanomat, 06/2021)
“… this new technology has enabled us to save many limbs” Prof. Piaggesi
Treating diabetic foot osteomyelitis without local antibiotics
As the world is searching for new solutions to combat infections due to antimicrobial resistance (AMR), Bonalive set out more than a decade ago to develop a novel concept to treat and prevent bone infections without the need of local antibiotics. The Bonalive S53P4 bioactive glass was developed as a bone regeneration solution and due to the proven bacterial growth inhibiting properties, the use has expanded to bone regeneration applications in septic bone surgery.
The first pilot study, with 11 patients, was published in 2010 and the CE mark for bone cavity filling in the treatment of chronic osteomyelitis was approved in 2011. Since then, 13 retrospective studies (376 patients), 3 prospective studies (37 patients) and 6 comparative studies with control groups (124 patients) have been published with our Bonalive products, as the first CE- marked solution in the world, to treat bone infections in orthopedics and traumatology without local antibiotics. See our most notable references here.

Mechanism of action
The basic antimicrobial mechanism of action is based on physical-chemical ion-exchange surface reactions that occur after implantation that result in a locally increased pH (pH 9-11) and increased osmotic pressure. The main benefit of using this mechanism, in contrast to the traditional means with antibiotics, is that no local antibiotics are needed, efficacy has been proven towards resistant bacteria (e.g., MRSE and MRSA) 7, 8, 9, it has a broad-spectrum efficacy towards both gram positive and gram-negative bacteria 10, 11, antibiofilm activity has been evidenced12 and the mechanism does not promote resistance selection13.
We have only started to understand what is happening at the interface of the technology (S53P4 bioactive glass) and biology (bone). As S53P4 is a bioactive biomaterial, a cascade of reactions is elicited that are thought to have an impact on multiple levels.
Properties of S53P4 bioactive glass:
– Bacterial growth inhibiting14–15
– osteostimulative16–17
Studies have also revealed the following properties:
– angiogenesis stimulation18–19
– anti-inflammatory and immuno-stimulatory properties20–21
All these properties of the researched S53P4 bioactive glass are thought to contribute to the healing process of these challenging patients.
How does bioactive glass help address these needs? Is bioactive glass a future option?
 The International Working Group on the Diabetic Foot has stated in their previous guideline published in 2019 that: “The increasing problem of infection with antibiotic resistant organisms demands development of alternative treatments to standard antibiotic therapy. Various types of antiseptics have been used to treat diabetic foot ulcers, but the available evidence does not support any beneficial effect for most of these”.
The International Working Group on the Diabetic Foot has stated in their previous guideline published in 2019 that: “The increasing problem of infection with antibiotic resistant organisms demands development of alternative treatments to standard antibiotic therapy. Various types of antiseptics have been used to treat diabetic foot ulcers, but the available evidence does not support any beneficial effect for most of these”.
The first clinical experiences showed a significantly higher infection resolution rate (90% vs 61.9%) compared to Standard of Care.
We are very inspired by the first clinical experiences in Italy with S53P4 bioactive glass (Bonalive® granules), as they showed a significantly higher infection resolution rate (90% vs 61.9%) compared to Standard of Care (SoC)22. This suggests we have an opportunity to generate a radical change in diabetic foot treatment if this hold true in the larger studies that we are running.
This concept we are building will be a major step towards sustainable healthcare, where infections are treated without antibiotics.
Dr. Fredrik Ollila
Founder and Chairman
Bonalive Biomaterials Ltd
If you are a physician and you would like to learn more about Bonalive® granules bioactive glass for diabetic foot osteomyelitis surgery, sign up to the Smart Healing Center for patient cases, publications, peer support and more.